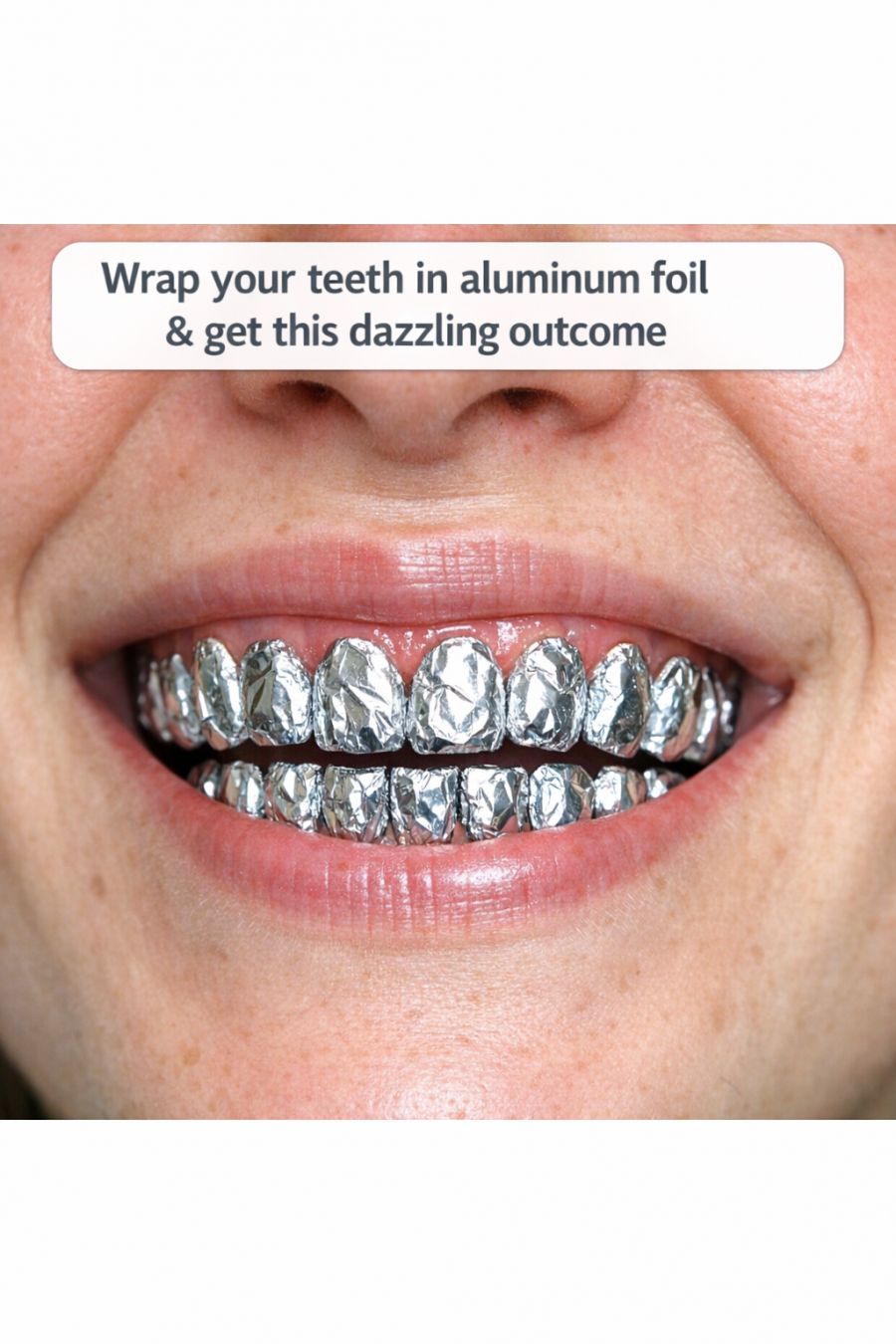
In the quest for a dazzling smile, people have turned to a myriad of remedies, both conventional and unconventional. One such trend that has captured the curiosity of many is the idea of wrapping teeth in aluminum foil, promising a quick, brightening effect. But what exactly does this process entail, and is it too good to be true?
The promise of whiter teeth in minutes with everyday household items is undeniably appealing. However, the safety and efficacy of such methods remain questionable. In this article, we explore the origins of this viral hack, the science behind it, and the real implications of using aluminum foil on your teeth.
1. What Really Happens When You Wrap Teeth in Aluminum Foil
The concept of wrapping teeth in aluminum foil typically involves covering the teeth with a layer of foil after applying a mixture of baking soda and toothpaste. The idea is that the foil acts as a conductor, amplifying the whitening effects of the paste. However, the actual impact is quite limited. Aluminum foil, measuring typically around 0.016 mm thick, is not designed to interact with the chemical components of toothpaste or baking soda in a way that would enhance whitening. Instead, the foil simply compresses the paste against the teeth without any additional chemical reaction.
2. The Viral Foil-Teeth Hack: Where Did This Trend Start?
The aluminum foil teeth whitening hack gained traction through various social media platforms, where DIY enthusiasts often share unconventional beauty tips. The trend seems to have originated from a series of viral videos and posts that showcased dramatic before-and-after transformations. These posts, often devoid of scientific backing, capitalize on the desire for instant results and the allure of using common household items for beauty enhancements.
3. The Science of Enamel, Heat, and Metal Contact
Enamel, the outermost layer of teeth, is composed of hydroxyapatite crystals and is the hardest substance in the human body. However, it is not impervious to damage, particularly from acidic or abrasive substances. The use of aluminum foil in contact with teeth introduces the potential for galvanic reactions, especially if the foil comes into contact with amalgam fillings, which could generate heat and cause discomfort. Additionally, the abrasive nature of baking soda can gradually wear down enamel if used excessively.
4. Potential Short-Term Effects: Sensations, Stains, and Shine
Some users report a temporary sensation of increased tooth sensitivity after using the foil method, likely due to the abrasion from baking soda. While there might be a perceived initial shine, this is often just the result of a thorough cleaning rather than any chemical whitening. Any apparent reduction in surface stains is usually superficial and does not equate to actual whitening.
5. The Hidden Risks: Enamel Damage, Sensitivity, and Burns
The repeated use of abrasive substances like baking soda can lead to enamel erosion over time, increasing the risk of cavities and tooth sensitivity. Moreover, if the aluminum foil method is combined with other acidic or abrasive DIY treatments, it could exacerbate these effects. Users with fillings, crowns, or other dental work may also experience galvanic reactions, leading to discomfort or even minor burns in the oral cavity.
continued on the next page